Home \ Knowledge Hub \ Focus on \ Focus On… A Decision Tool for FMD

01 Apr 2016
Focus On… A Decision Tool for FMD
SHARE

ALASDAIR KING
Executive Director, International Veterinary Health
Introducing a Decision Tool
While a lot of information on FMD control focusses on the epidemic situation, there is less information available for endemic countries to make strategic decisions to control the disease. Key factors such as the correct use of sometimes limited resources and long-term planning instead of short-term reactions are important to consider. Working with LEI Wageningen University, we have developed an interactive modelling tool that can provide governments with important information on the economic impact of different strategies. This tool has been presented at a number of FMD conferences, and in this edition of “Focus On…” we will provide you with further details.
Data Entry
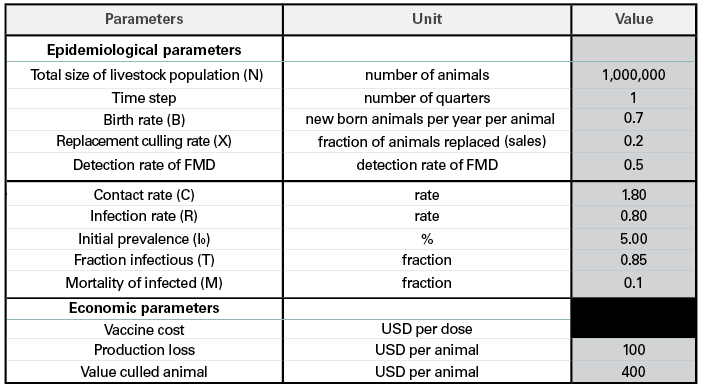
The first step to using the tool is to enter country-specific parameters. These include the livestock population, disease prevalence, mortality rates, and the costs involved in production loss (Figure 1). The better the information available, the better the end report will be, and surveillance is important to fully understand the disease situation in the country. However, if the information is not available, estimates can be used.
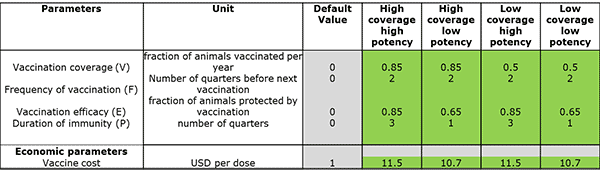
Having defined the situation in the country, different vaccination scenarios can be entered. In the example in Figure 2, we have entered combinations of high vs low coverage and high vs low potency. We have assumed that the vaccination campaign will cost $10/animal, and that a low-potency vaccine costs $0.70/dose, compared to $1.50/dose for a high-potency vaccine. These numbers for vaccine performance are consistent with other research, but we have exaggerated the difference in price between the two types of vaccine. It is important to remember that individual countries can adjust these figures to suit their own situations.
Results
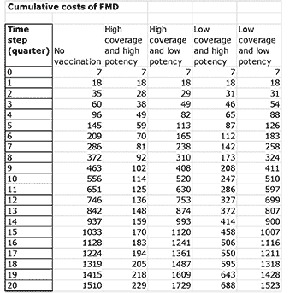
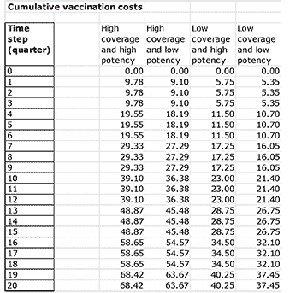
Once all the parameters are entered, the model can be run. This generates the data and graphs for analysis. For instance, Figure 3 demonstrates the cumulative costs of disease and vaccination over time.
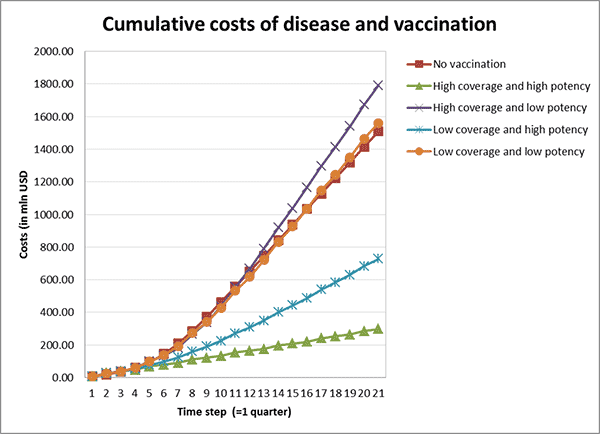
The value of visually representing the impact of different strategies is evident when looking at the cumulative costs. Often, when developing strategies, the procurement process can focus on the cost of the vaccine. In addition, when only considering the next campaign, or quarter, then the cost of a high-potency vaccine can appear too large, leading to cost-cutting decisions. However, it can be seen that the high-coverage, high-potency strategy is easily the most cost effective over time. This is because the costs of the disease itself going unchecked massively outweigh any potential cost of vaccine (see Figures 4 and 5).
A Dynamic Approach
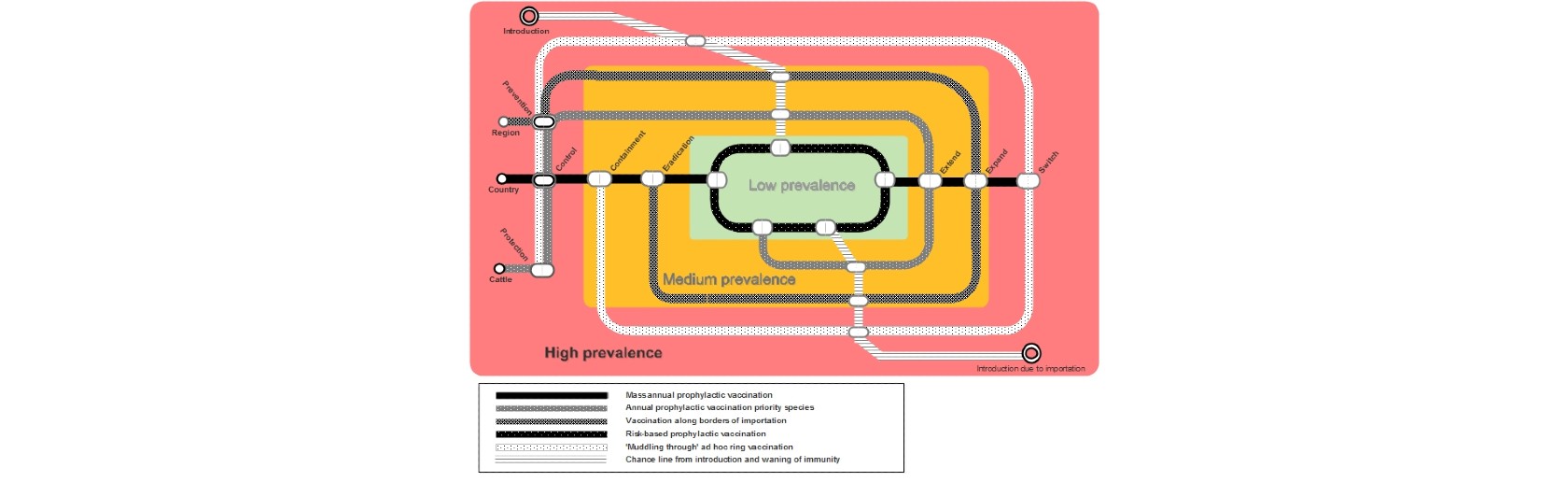
FMD strategies need to be dynamic. A single strategy will not work over a long period, and it is therefore important to adapt to changes in conditions over time. The modelling tool can be applied to a dynamic adaptive pathway approach, wherein the FMD situation is seen as a “subway map” (see Figure 6).
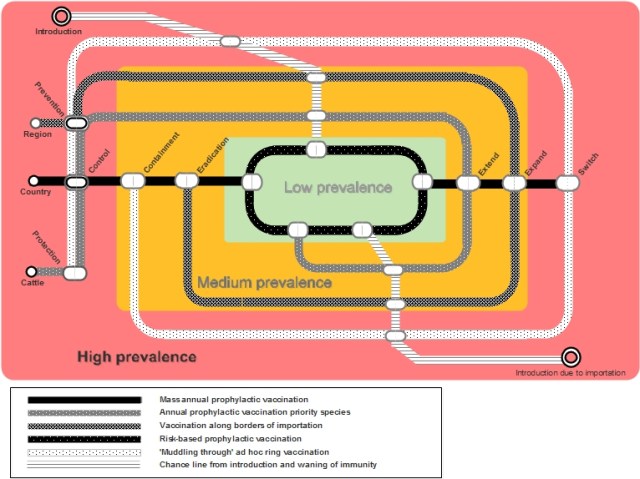
This approach allows policy makers to identify the possibilities to switch from one option to another. A number of considerations are required in order to best determine when and where to change strategy, such as the following:
- The uncertainties about initial FMD prevalence
- The uncertainties about contract rate or infection rate between herds
- Practical constraints
- Improved surveillance
- Changes in field strains of virus
Subsequently, possible switching points are explored by running the simulation model with varying parameters. This ‘what-if’ type of analysis can be used to assess the uncertainties of future FMD situations. Results of the ‘what-if’ analysis provide insight into the possible leeway in implementing different vaccination campaigns.
Supporting Strategy and Planning
The goal of this research is to support decision-making with regard to vaccination campaigns in an endemic situation by highlighting the options and the considerations. For decision support, it is necessary to factor in country objectives when assessing the socioeconomic consequences of FMD and FMD intervention strategies. Choosing the right options at the right time should be based on practical resource constraints and uncertainties about FMD prevalence and spread. This tool can help governments make informed decisions.
Thanks
With thanks to Dr. Lan Ge, Dr. Marcel van Asseldonk, and Dr. Ron Bergevoet of LEI Wageningen UR for developing the model for this work.
References
Choi, Y.K., Johnson, W.O., Jones, G., Perez, A., & Thurmond, M.C. (2012). Modelling and predicting temporal frequency of foot-and-mouth disease cases in countries with endemic foot-and-mouth disease. Journal of the Royal Statistical Society. Series A: Statistics in Society, 175(2), 619-636.
Elnekave, E., Li, Y., Zamir, L., Even-Tov, B., Hamblin, P., Gelman, B., et al. (2013). The field effectiveness of routine and emergency vaccination with an inactivated vaccine against foot and mouth disease. Vaccine, 31(6), 879-885.
Ge, L., Mourits, M. C. M., & Huirne, R. B. M. (2007). Towards flexible decision support in the control of animal epidemics. Scientific and Technical Review of the OIE, 26(3), 551-563.
Haasnoot, M., Kwakkel, J.H., Walker, W.E., & ter Maat, J. (2013). Dynamic adaptive policy pathways: A method for crafting robust decisions for a deeply uncertain world. 23(2), 485-498.
Jemberu, W.T., Mourits, M.C.M., Woldehanna, T., & Hogeveen, H. (2014). Economic impact of foot and mouth disease outbreaks on smallholder farmers in Ethiopia. Preventive Veterinary Medicine, 116(1-2), 26-36.
Kitching, P., Hammond, J., Jeggo, M., Charleston, B., Paton, D., Rodriguez, L., et al. (2007). Global FMD control-Is it an option? Vaccine, 25(30 SPEC. ISS.), 5660-5664.
Knight-Jones, T.J.D., & Rushton, J. (2013). The economic impacts of foot and mouth disease – What are they, how big are they and where do they occur? Preventive Veterinary Medicine, 112(3-4), 162-173.
Leforban, Y. and K. Sumption, 2010, Foot and mouth disease, in Legevre et al. ed. Infectious and Parasitic Diseases of Livestock, pp. 299-324, Lavoisier, Paris.
Mahul, O., & Gohin, A. (1999). Irreversible decision making in contagious animal disease control under uncertainty: an illustration using FMD in Brittany. European Review of Agricultural Economics, 26(1), 39-58.
Parida, S. (2009). Vaccination against foot-and-mouth disease virus: Strategies and effectiveness. Expert Review of Vaccines, 8(3), 347-365.
Rodriguez, L.L., & Gay, C.G. (2011). Development of vaccines toward the global control and eradication of foot-and-mouth disease. Expert Review of Vaccines, 10(3), 377-387.
Roth F., Zinsstag J., Orkhon D., Chimed-Ochir G., Hutton G., Cosivi O., Carrin G. & Otte J. (2003). Human health benefits from livestock vaccination for brucellosis: case study. Bull. WHO, 81, 867–876.
Rushton, J. (2008). Economic aspects of foot and mouth disease in Bolivia. OIE Revue Scientifique et Technique, 27(3), 759-769.
Rweyemamu, M., Roeder, P., MacKay, D., Sumption, K., Brownlie, J., & Leforban, Y. (2008). Planning for the progressive control of foot-and-mouth disease worldwide. Transboundary and Emerging Diseases, 55(1), 73-87.
Zinsstag J. (2007). Human benefits of animal interventions for zoonosis control. Emerging Infectious Diseases, 13, 527-531.
ALASDAIR KING
Executive Director, International Veterinary Health

