Home \ Knowledge Hub \ Focus on \ Focus On… Life as a Vet in Research – Transboundary and Emerging Diseases


10 May 2020
Focus On… Life as a Vet in Research – Transboundary and Emerging Diseases
SHARE

BEATRIZ SANZ-BERNARDO
The Pirbright Institute
A bold move from practice to research
Almost 15 years ago, when I graduated as a veterinary surgeon, I never envisioned that I would be researching within infectious diseases… I was going to be a large animal practitioner! I guess I started to take this path as a newly graduated vet. While I was doing my clinical training in livestock medicine and surgery, my interest in infectious diseases started to grow while working with dairy calves. I came across so many reported risk factors associated with disease outcome, yet still the individual animal seemed to play quite a big role in the outcome of disease. I wanted more answers, I wanted to know more, but I needed to be able to understand the research done at a level below the individual animal.
My interest in infectious diseases started to grow as a newly graduated livestock vet. I wanted answers, I wanted to know more, and I needed to understand the research below the individual animal.
That is why I decided to continue my education in molecular research with a focus in virology and immunology. This was a bold move because I loved so much doing surgeries, diagnosing and treating animals, being all day in the field… and all the lab experience that I had by that time was a few courses during the veterinary and master’s degrees. The move has been very rewarding though, not only have I learned to interpret and use techniques commonly employed to study viral pathogenesis, but I have also been working during these years with very interesting viral diseases of high consequences: peste des petits ruminants (PPR), foot and mouth disease (FMD), bluetongue and more recently with lumpy skin disease (LSD).
Lumpy skin disease
LSD was first described 90 years ago and was restricted within the African continent. As with many other endemic diseases, African farmers have dealt with the economic impact of the disease due to loss of production, treatment and vaccination costs. In recent years, this disease has demonstrated a similar pattern to many other vector-borne diseases by expanding its geographical location to new areas and becoming a threat for disease-free countries that need evidence-based control strategies to stop the spread of the virus. Vaccination is available for the control of LSD; but there are still knowledge gaps in the epidemiology, pathogenesis and diagnostics which could be improved to help in the implementation of comprehensive effective control programs.
In recent years, LSD has demonstrated a similar pattern to many other vector-borne diseases by expanding its geographical location to new areas and becoming a threat for disease-free countries. Vaccination is effective to control LSD but there are still many knowledge gaps in epidemiology.
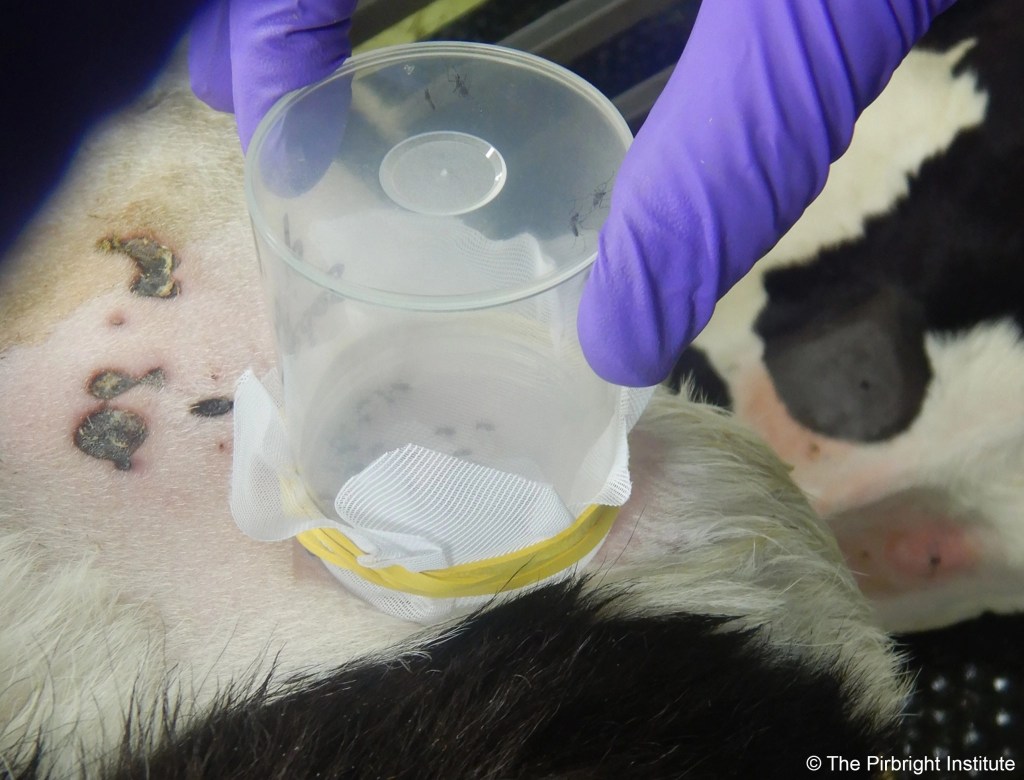
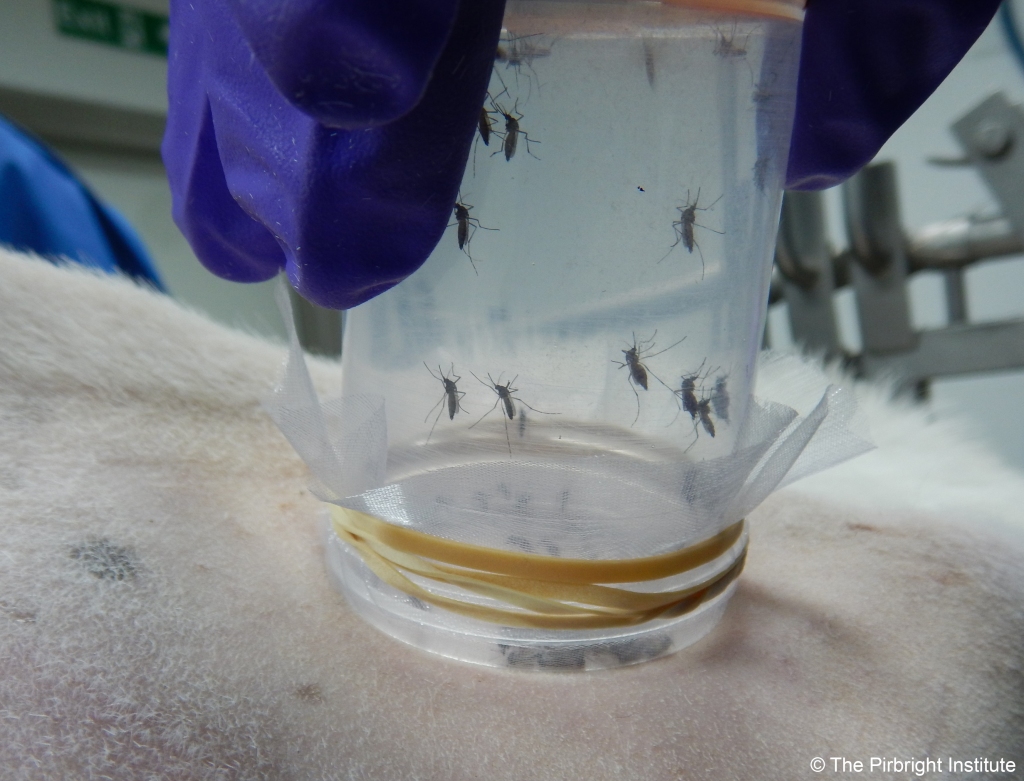
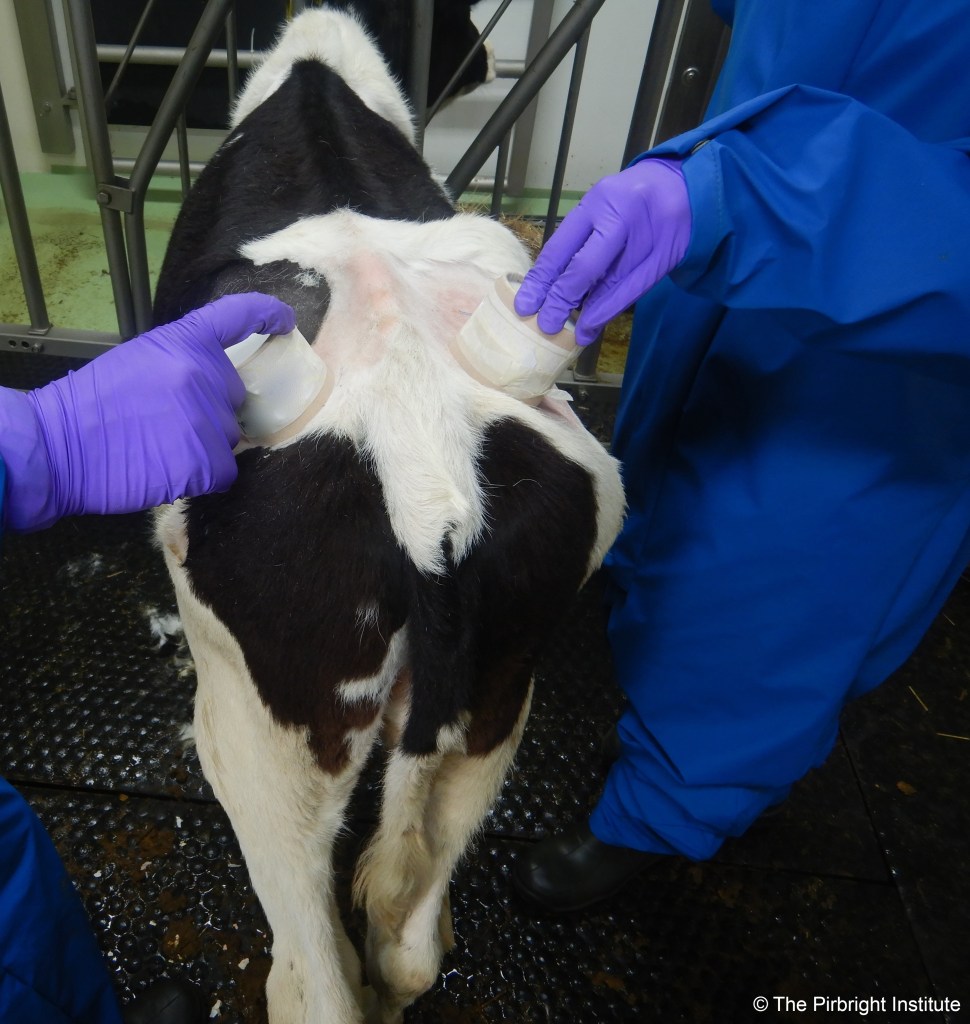
My personal point of view is that it is very important that we advance the identification of those animals naturally challenged by LSDV which do not develop the characteristic systemic disease with cutaneous nodules. We need to understand the epidemiology of this population and their relevance for transmission. In order to play a role, and for transmission to occur, live virus must be accessible in these non-clinical animals to the vectors (either in the blood and/or skin). If they are actually a population of truly resistant animals (animals which have cleared the infection upon challenge), all efforts should be established to detect and maintain them in our herds, as a natural measure of control for LSDV transmission. However, it is early days to make big claims. Before we can take steps to establish the role of such animals in the epidemiology of LSDV, there is a question in need of answering: what tools do we have to identify asymptomatic/subclinical animals (infection with few or no clinical signs) or resistant animals to LSDV?
LSD is characterised by a relatively low and short viremia in clinical animals, which is very difficult to detect in animals not developing the clinical disease as they have much lower levels of circulating virus. We will need to improve diagnostic techniques combined with suitable surveillance if we want to accurately estimate the prevalence of subclinical cases by detection of the virus. Another surveillance technique is sero-surveillance, but current methodology detecting capripoxvirus-specific antibodies cross-reacts with all three species (sheep-poxvirus and goat-poxvirus (SGPV) or LSDV). We know that cattle generate these antibodies when exposed to either SGPV or LSDV, so when mixed infections are in the area of surveillance this method could overestimate the number of subclinical animals.
Working within Pip Beard’s lab at The Pirbright Institute, we are trying to obtain stronger evidence of the importance of different dipteran vectors in LSDV transmission, but we have also explored the effect of animals without clinical signs after challenge in the overall transmission of the virus. We will be publishing details of our findings shortly (see below), and much more research on LSDV is being carried currently which will help to further improve our understanding of the disease and control strategies.
Lumpy Skin Disease Is Characterized by Severe Multifocal Dermatitis With Necrotizing Fibrinoid Vasculitis Following Experimental Infection. Veterinary Pathology 57 (3) , 388-396: https://doi.org/10.1177%2F0300985820913268
Full time research is something that many vet students don’t consider seriously, and I was one of them. Now I am very glad that I decided to explore more in detail various aspects of livestock diseases, and although I can’t say that I don’t miss performing surgeries, I am still fulfilling the remit of the veterinary profession and my career goals.


BEATRIZ SANZ-BERNARDO
The Pirbright Institute
“The views expressed in this article are those of the author(s) and do not necessarily represent those of
MSD Animal Health.”

